By PHILIP MARCELO
NEW YORK (AP) — Nude photos. The names and faces of sexual abuse victims. Bank account and Social Security numbers in full view.
Related Articles
Plastic surgeons group calls for delaying gender-affirming surgery until age 19
Melania Trump meets with freed American-Israeli hostage whose story she features in her new film
Former Indiana Rep. Lee Hamilton, leader of Sept. 11 panel, dies
Supreme Court allows new California congressional districts that favor Democrats
Government lawyer yanked from immigration detail in Minnesota after telling judge ‘this job sucks’
All of these things appeared in the mountain of documents released publicly by the U.S. Justice Department as part of its effort to comply with a law requiring it to open its investigative files on Jeffrey Epstein.
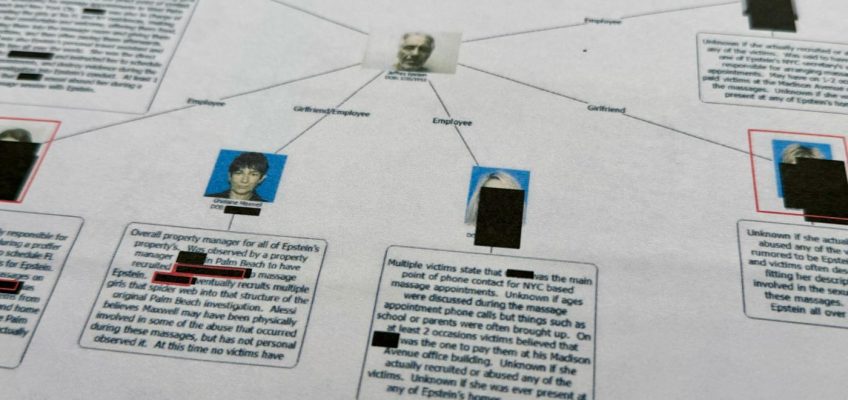
That law was intended to preserve important privacy protections for Epstein’s victims. Their names were supposed to have been blacked out in documents. Their faces and bodies were supposed to be obscured in photos.
Mistakes, though, have been rampant. A review by The Associated Press and other news organizations has found countless examples of sloppy, inconsistent or nonexistent redactions that have revealed sensitive private information.
A photo of one girl who was underage when she was hired to give sexualized massages to Epstein in Florida appeared in a chart of his alleged victims. Police reports with the names of several of his victims, including some who have never stepped forward to identify themselves publicly, were released with no redactions at all.
Despite the Justice Department’s efforts to fix the oversights, a photo of one topless woman remained on the site, with her face in full view, Wednesday evening.
Some accusers and their lawyers called this week for the Justice Department to take down the site and appoint an independent monitor to prevent further errors.
A judge scheduled a hearing for Wednesday in New York on the matter, then cancelled it after one of the lawyers for victims cited progress in resolving the issues. But that lawyer, Brittany Henderson, said they were still weighing “all potential avenues of recourse” to address the “permanent and irreparable” harm caused to some women.
“The failure here is not merely technical,” she said in a statement Wednesday. “It is a failure to safeguard human beings who were promised protection by our government. Until every document is properly redacted, that failure is ongoing.”
Annie Farmer, who said she was 16 when she was sexually assaulted by Epstein and his confidante, Ghislaine Maxwell, said that while her name has previously been public, other details she’d rather be kept private, including her date of birth and phone number, were wrongly revealed in the documents.
“At this point, I’m feeling really most of all angry about the way that this unfolded,” she told NBC News. “The fact that it’s been done in such a beyond careless way, where people have been endangered because of it, is really horrifying.”
Trump administration defends its Epstein files redaction efforts
The Justice Department has blamed technical or human errors on the problems and said it has taken down many of the problematic materials and is working to republish properly redacted versions.
The task of reviewing and blacking out millions of pages of records took place in a compressed time frame. President Donald Trump signed the law requiring the disclosure of the documents on Nov. 19. That law gave the Justice Department just 30 days to release the files. It missed that deadline, in part because it said it needed more time to comply with privacy protections.
Hundreds of lawyers were pulled from their regular duties, including overseeing criminal cases, to try and complete the document review — to the point where at least one judge in New York complained that it was holding up other matters.
The database, which is posted on the Justice Department website, represents the largest release of files to date in the yearslong investigations into Epstein, who killed himself in a New York jail cell in 2019 while awaiting trial on federal sex trafficking charges.
Epstein files rife with missed or incomplete redactions
Associated Press reporters analyzing the documents have so far found multiple examples of names and other personal information of potential victims revealed.
They have also found many cases of overzealous redactions.
In one news clipping included in the file, the Justice Department apparently blacked out the name “Joseph” from a photo caption describing a nativity scene at a California church. “A nativity scene depicting Jesus, Mary and (REDACTED),” it said.
In an email released in the files, a dog’s name appeared to have been redacted: “I spent an hour walking (REDACTED) and then another hour bathing her blow drying her and brushing her. I hope she smells better!!” the email said.
The Justice Department has said staff tasked with preparing the files for release were instructed to limit redactions only to information related to victims and their families, though in many documents the names of many other people were blacked out, including lawyers and public figures.
Images remain uncensored
The Justice Department has said it intended to black out any portion of a photo showing nudity, and any photos of women that could potentially show a victim.
In some photos reviewed by The AP, those redactions did obscure women’s faces, but left plenty of their bare skin exposed in a way that would likely embarrass the women anyway. Photos showed identifiable women trying on outfits in clothing store dressing rooms or lounging in bathing suits.
One set of more than 100 images of a young woman were nearly all blacked out, save for the very last image, which revealed her entire face.
Associated Press reporters from around the world contributed to this report.
The AP is reviewing the documents released by the Justice Department in collaboration with journalists from CBS, NBC, MS NOW and CNBC. Journalists from each newsroom are working together to examine the files and share information about what is in them. Each outlet is responsible for its own independent news coverage of the documents.